Your views on the future of Personalised Medicine and increasing engagement
The aim of our study is to bring personalised medicine for people with Crohn’s and Colitis one step closer. Personalised medicine is when treatment is tailored to individuals.
There is currently no cure for Crohn’s or Colitis. People living with the conditions experience unpredictable periods of relapse, known as ‘flare-ups’, and remission. Medicines can help in many cases, but not all. Some people do not respond to any medicines, and surgery may be needed to remove inflamed bowel sections. All this can have a big impact on people’s quality of life, mental health, and wellbeing.
Recommended treatments include medicines called ‘biologics’. Biologics aim to get symptoms under control and keep them under control. These medicines work by blocking the proteins, called cytokines, that ‘switch-on’ inflammatory responses. This, in turn, reduces inflammation. Biologics are now the most prescribed treatment for Ulcerative Colitis. There are many different types, each targeting a different inflammatory protein. For some people, a particular biologic may help reduce inflammation and control symptoms, but for others it does not. Some people need to try several biologics before they find one that works for them.
Our research is being shaped by people with lived experience (both Crohn’s and Colitis) and will investigate bowel inflammation at a microscopic and molecular level. Specifically, the project will explore ways to match individuals with Ulcerative Colitis to the medicine that will work best for them. This will mean that their symptoms are controlled more quickly and they have a better quality of life.
When we recently used a microscope to look at tissue samples taken from people with Ulcerative Colitis, we found that different cells and molecules contribute to disease in different people, so bowel inflammation is individual to each person. Despite these differences in the features of the disease, biologics are prescribed in a ‘one-size-fits-all’ way. This approach does not consider the individual molecular characteristics of the person’s bowel inflammation at the time of treatment. By targeting treatment according to the individual’s molecular pattern, our research aims to decrease the failure rates of current treatments and bring us much closer to personalised medicine for Ulcerative Colitis.
In this focus group, we asked participants for thoughts on whether you would be willing to try a personalised medicines approach – if proven successful first – in a clinical trial setting. We would also discussed ideas on how patient engagement groups can get more involved in research.
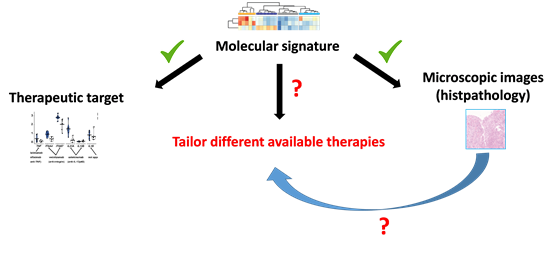
Schematic overview: Decades of research have helped us to find molecules that can be targeted by medications to stop or reduce intestinal inflammation. Similarly we recently found that that certain molecular signatures of inflammation are reflected on microscopic images already taken for diagnostic purposes in hospitals. The studies we are conducting together with patient engagement and Crohn’s & Colitis UK are aiming to find out whether either molecular signatures or microscopic images – or both – can be used to better tailor medicines to individuals.